COVID-19 control with a larger NPI space
multiple_NPIs.RmdOverview
This vignette demonstrates model predictive optimal control of ICU cases for COVID-19
Load Libraries and Setup
Load the required libraries and initialize the simulation environment.
library(EpiControl)
library(VGAM)
library(parallel)
library(pbapply)
library(zoo) # For rolling sum operations
library(dplyr)
library(ggplot2)
set.seed(1)Initialize the cluster for parallel computing:
cores <- detectCores() - 1
cl <- makeCluster(cores)
clusterSetRNGStream(cl, iseed = 20250501)Epidemiological and Noise Parameters
Define parameters for pathogens and noise: Can include different pathogens, in this case we consider COVID-19 and Ebola virus.
Epi_pars <- data.frame(
Pathogen = c("COVID-19", "Ebola"),
R0 = c(3.5, 2.5),
gen_time = c(6.5, 15.0),
gen_time_var = c(2.1, 2.1),
CFR = c(0.0132, 0.5),
mortality_mean = c(10.0, 10.0),
mortality_var = c(1.1, 1.1)
)
Noise_pars <- data.frame(
repd_mean = 10.5, # Reporting delay mean
del_disp = 5.0, # Reporting delay variance
ur_mean = 0.3, # Under-reporting mean
ur_beta_a = 50.0 # Beta distribution alpha for under-reporting
)Action Space for Policy Interventions
Define non-pharmaceutical interventions: We have 2 actions, introducing a “lockdown” and no interventions.
# we can use 3, 6 or 10 levels
#Action_space <- data.frame (
# NPI = c("No restrictions", "Social distancing", "Lockdown"),
# R_coeff = c(1.0, 0.5, 0.2), #R0_act = R0 * ctrl_states
# R_beta_a = c(0.0, 5.0, 5.0), #R0_act uncertainty
# cost_of_NPI = c(0.0, 0.01, 0.15)
#)
#Action_space <- data.frame (
# NPI = c("No restrictions", "SD1", "SD2", "SD3", "SD4", "Lockdown"),
# R_coeff = c(1.0, 0.8, 0.6, 0.4, 0.2, 0.1), #R0_act = R0 * ctrl_states
# R_beta_a = c(0.0, 5.0, 5.0, 5.0, 5.0, 5.0), #R0_act uncertainty
# cost_of_NPI = c(0.0, 0.02, 0.05, 0.08, 0.15, 0.35)
#)
Action_space <- data.frame (
NPI = c("No restrictions", "SD1", "SD2", "SD3", "SD4", "SD5", "SD6", "SD7", "SD8", "Lockdown"),
R_coeff = c(1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, 0.1), #R0_act = R0 * ctrl_states
R_beta_a = c(0.0, 5.0, 5.0, 5.0, 5.0, 5.0, 5.0, 5.0, 5.0, 5.0), #R0_act uncertainty
cost_of_NPI = c(0.0, 0.01, 0.02, 0.03, 0.04, 0.05, 0.065, 0.08, 0.15, 0.35)
)Simulation Setup
Set up key parameters and initialize simulation data:
C_target <- 5000
D_target <- 1
r_trans_len <- 7
sim_settings <- list(
ndays = 121 * 7, #simulation length
start_day = 1,
N = 3.5e6, # population size
I0 = 10, # initial infections
C_target = C_target, #target cases
C_target_pen = C_target*1.5, #overshoot penalty threshold
R_target = 1.0,
D_target = D_target, #one way to get peaks at 400 is to increase this to 15
D_target_pen = 50, #max death
alpha = 1.3/C_target, #~proportional gain (regulates error in cases) covid
#alpha = 3.25/C_target #~proportional gain (regulates error in cases) ebola
alpha_d = 0*1.3/D_target,
ovp = 5.0, #overshoot penalty
dovp = 0*10.0, #death overshoot penalty
gamma = 0.95, #discounting factor
n_ens = 20L, #MC assembly size for 4
sim_ens = 20L, #assembly size for full simulation
rf = 7L, #days 14
R_est_wind = 5L, #rf-2 #window for R estimation
pred_days = 12L,
r_trans_steep = 1.5, # Growth rate
r_trans_len = r_trans_len, # Number of days for the transition
t0 = r_trans_len / 2, # Midpoint of the transition
pathogen = 1,
susceptibles = 1,
delay = 0,
ur = 0,
r_dir = 2
)
column_names <- c("days", "sim_id", "I", "Lambda", "C", "Lambda_C", "S", "Deaths", "Re", "Rew", "Rest", "R0est", "policy", "R_coeff")
episim_data <- data.frame(matrix(0, nrow = (sim_settings$ndays), ncol = length(column_names)))
colnames(episim_data) <- column_names
episim_data$policy <- rep(1, sim_settings$ndays)
episim_data$days <- 1:(sim_settings$ndays)
episim_data[1, ] <- c(1, 1, sim_settings$I0, sim_settings$I0, Noise_pars$ur_mean * sim_settings$I0, Noise_pars$ur_mean * sim_settings$I0, sim_settings$N - sim_settings$I0, 0, Epi_pars[1, "R0"], Epi_pars[1, "R0"], 1, 1, 1, 1)
episim_data_ens <- replicate(sim_settings$sim_ens, episim_data, simplify = FALSE)
for (ii in 1:sim_settings$sim_ens) {
episim_data_ens[[ii]]$sim_id <- rep(ii, sim_settings$ndays)
}Running the Simulation
Run simulations in parallel using the pblapply function:
episettings <- list(
sim_function = Epi_MPC_run_wd,
reward_function = reward_fun_wd,
R_estimator = R_estim,
noise_par = Noise_pars,
epi_par = Epi_pars,
actions = Action_space,
sim_settings = sim_settings,
parallel = TRUE
)
episettings$cl <- cl
results <- epicontrol(episim_data_ens, episettings)
#> [1] 1
#> | | | 0%
episim_data_ens <- results
stopCluster(cl)
#for (jj in 1:sim_ens) {
# episim_data_ens[[jj]] <- Epi_MPC_run_wd(episim_data_ens[[jj]], Epi_pars, Noise_pars, Action_space, pred_days = pred_days, n_ens = n_ens, ndays = nrow(episim_data), R_est_wind = R_est_wind, pathogen = 1, susceptibles = 0, delay = 0, ur = 0, r_dir = 2, N = N)
# setTxtProgressBar(pb,jj)
#}
#close(pb)
for (jj in 1:sim_settings$sim_ens) {
episim_data_ens[[jj]] <- head(episim_data_ens[[jj]], -sim_settings$pred_days)
episim_data_ens[[jj]]["D_roll"] <- rollsum(episim_data_ens[[jj]]["Deaths"], 7, fill = NA)
episim_data_ens[[jj]]["I_roll"] <- rollsum(episim_data_ens[[jj]]["I"], 7, fill = NA)
episim_data_ens[[jj]]["D_cum"] <- cumsum(episim_data_ens[[jj]]["Deaths"])
episim_data_ens[[jj]]["I_cum"] <- cumsum(episim_data_ens[[jj]]["I"])
}
# Combine Simulation Results
combined_data <- do.call(rbind, episim_data_ens)Plotting results
combined_data <- combined_data %>%
group_by(sim_id, policy) %>%
arrange(days) %>%
mutate(group = cumsum(c(1, diff(days) != 1))) %>%
ungroup()
combined_data <- combined_data %>%
arrange(sim_id, days, policy)
policy_labels <- c("1" = "No restrictions", "2" = "SD1", "3" = "SD2", "4" = "SD3", "5" = "SD4", "6" = "SD5", "7" = "SD6", "8" = "SD7")
# find the day when herd immunity reached
# Sample constant value
HI <- 1/Epi_pars[1,"R0"]
# Assuming episim_data_ens[[1]]["S"] is a dataframe or list
# Extract the vector
S_vector <- episim_data_ens[[1]]["S"][[1]]
# Compute the fraction S/N
fraction <- S_vector / sim_settings$N
# Find the index where the fraction is first smaller than the constant
index <- which(fraction < HI)[1]
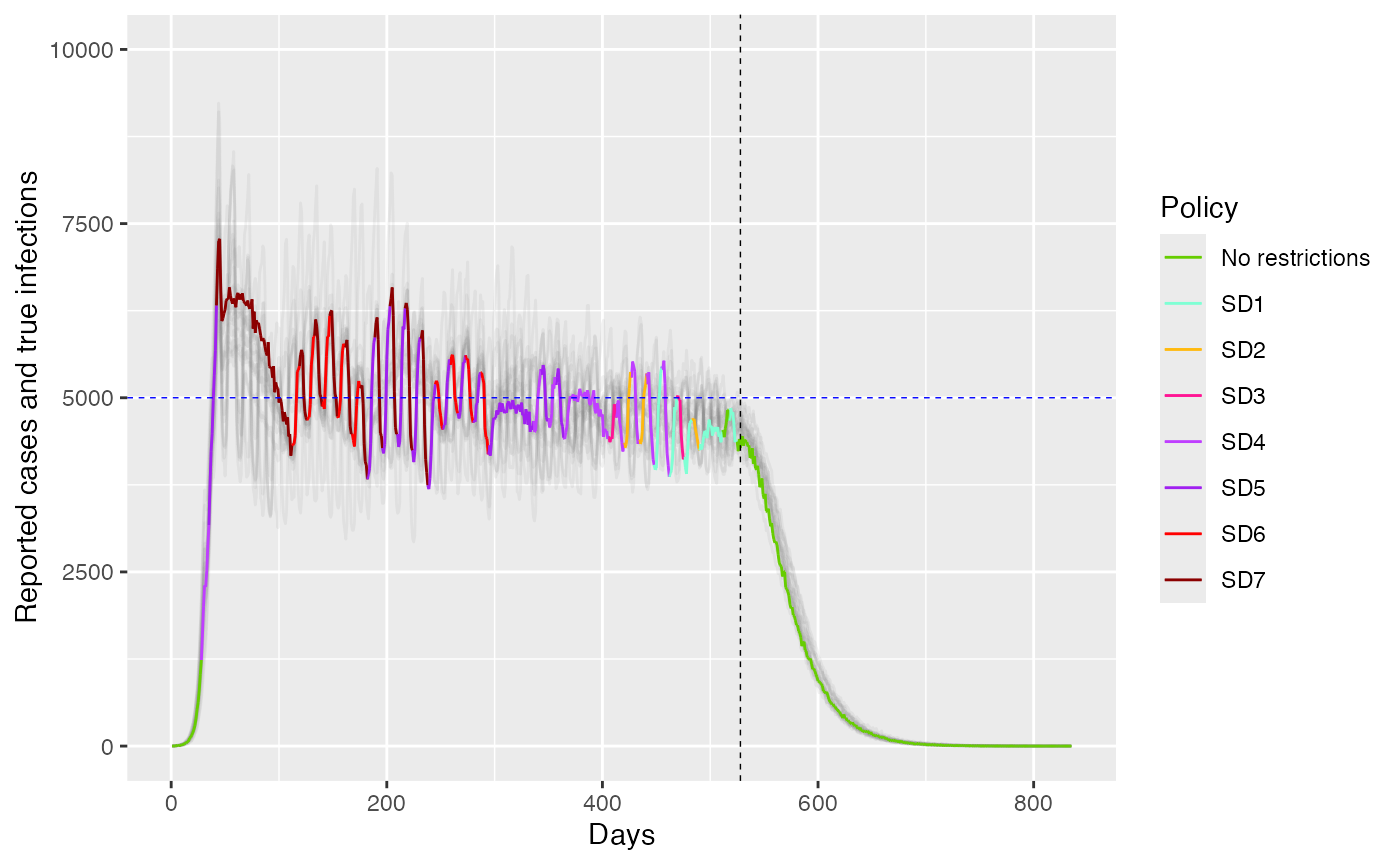
ggplot(combined_data %>% filter(sim_id == 1)) +
geom_line(data = subset(combined_data, sim_id != 1), aes(x = days, y = C, color = as.factor(sim_id)), alpha = 0.1) +
geom_line(aes(x = days, y = C, color = factor(policy, labels = policy_labels), group = 1), alpha = 1.0) +
geom_line(aes(x = days, y = I, color = factor(policy, labels = policy_labels), group = 1), alpha = 1.0, size=0.25) +
geom_hline(yintercept = C_target, linetype = "dashed", color = "blue", size=0.25) +
geom_vline(xintercept = index, linetype = "dashed", color = "black", size=0.25) +
labs(x = "Days", y = "Reported cases and true infections", color = "Policy") +
scale_color_manual(values = c("SD1" = "aquamarine", "SD2" = "darkgoldenrod1", "SD3" = "deeppink", "SD4" = "darkorchid1", "SD5" = "purple","SD6" = "red","SD7" = "darkred", "No restrictions" = "chartreuse3")) +
#scale_color_manual(values = c("SD1" = "aquamarine", "SD2" = "darkgoldenrod1", "SD3" = "deeppink", "SD4" = "darkorchid1", "SD5" = "purple", "No restrictions" = "chartreuse3")) +
guides(color = guide_legend(title = "Policy"))+
ylim(0,10000)